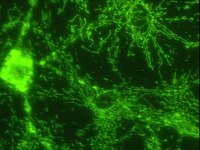
Endothelin-1 induces LIMK2-mediated programmed necrotic neuronal death independent of NOS activity.
Ko, AR; Hyun, HW; Min, SJ; Kim, JE; Kang, TC
Molecular brain
8
58
2015
Show Abstract
Recently, we have reported that LIM kinase 2 (LIMK2) involves programmed necrotic neuronal deaths induced by aberrant cyclin D1 expression following status epilepticus (SE). Up-regulation of LIMK2 expression induces neuronal necrosis by impairment of dynamin-related protein 1 (DRP1)-mediated mitochondrial fission. However, we could not elucidate the upstream effecter for LIMK2-mediated neuronal death. Thus, we investigated the role of endothelin-1 (ET-1) in LIMK2-mediated neuronal necrosis, since ET-1 involves neuronal death via various pathways.Following SE, ET-1 concentration and its mRNA were significantly increased in the hippocampus with up-regulation of ETB receptor expression. BQ788 (an ETB receptor antagonist) effectively attenuated SE-induced neuronal damage as well as reduction in LIMK2 mRNA/protein expression. In addition, BQ788 alleviated up-regulation of Rho kinase 1 (ROCK1) expression and impairment of DRP1-mediated mitochondrial fission in CA1 neurons following SE. BQ788 also attenuated neuronal death and up-regulation of LIMK2 expression induced by exogenous ET-1 injection.These findings suggest that ET-1 may be one of the upstream effectors for programmed neuronal necrosis through abnormal LIMK2 over-expression by ROCK1. | 26438559
 |
Cytomegalovirus-induced embryopathology: mouse submandibular salivary gland epithelial-mesenchymal ontogeny as a model.
Melnick, M; Mocarski, ES; Abichaker, G; Huang, J; Jaskoll, T
BMC developmental biology
6
42
2006
Show Abstract
Human studies suggest, and mouse models clearly demonstrate, that cytomegalovirus (CMV) is dysmorphic to early organ and tissue development. CMV has a particular tropism for embryonic salivary gland and other head mesenchyme. CMV has evolved to co-opt cell signaling networks so to optimize replication and survival, to the detriment of infected tissues. It has been postulated that mesenchymal infection is the critical step in disrupting organogenesis. If so, organogenesis dependent on epithelial-mesenchymal interactions would be particularly vulnerable. In this study, we chose to model the vulnerability by investigating the cell and molecular pathogenesis of CMV infected mouse embryonic submandibular salivary glands (SMGs).We infected E15 SMG explants with mouse CMV (mCMV). Active infection for up to 12 days in vitro results in a remarkable cell and molecular pathology characterized by atypical ductal epithelial hyperplasia, apparent epitheliomesenchymal transformation, oncocytic-like stromal metaplasia, beta-catenin nuclear localization, and upregulation of Nfkb2, Relb, Il6, Stat3, and Cox2. Rescue with an antiviral nucleoside analogue indicates that mCMV replication is necessary to initiate and maintain SMG dysmorphogenesis.mCMV infection of embryonic mouse explants results in dysplasia, metaplasia, and, possibly, anaplasia. The molecular pathogenesis appears to center around the activation of canonical and, perhaps more importantly, noncanonical NFkappaB. Further, COX-2 and IL-6 are important downstream effectors of embryopathology. At the cellular level, there appears to be a consequential interplay between the transformed SMG cells and the surrounding extracellular matrix, resulting in the nuclear translocation of beta-catenin. From these studies, a tentative framework has emerged within which additional studies may be planned and performed. | 16959038
 |
Proteomic identification of the involvement of the mitochondrial rieske protein in epilepsy.
Heike Junker, Kira Späte, Yalikun Suofu, Reinhard Walther, Gerold Schwarz, Winfried Kammer, Alfred Nordheim, Lary C Walker, Uwe Runge, Christof Kessler, Aurel Popa-Wagner
Epilepsia
46
339-43
2005
Show Abstract
PURPOSE: Kindled seizures are widely used to model epileptogenesis, but the molecular mechanisms underlying the attainment of kindling status are largely unknown. Recently we showed that achievement of kindling status in the Sprague-Dawley rat is associated with a critical developmental interval of 25 +/- 1 days; the identification of this long, well-defined developmental interval for inducing kindling status makes possible a dissection of the cellular and genetic events underlying this phenomenon and its relation to normal and pathologic brain function. METHODS: By using proteomics on cerebral tissue from our new rat kindling model, we undertook a global analysis of protein expression in kindled animals. Some of the identified proteins were further investigated by using immunohistochemistry. RESULTS: We report the identification of a modified variant of the Rieske iron-sulfur protein, a component of the mitochondrial cytochrome bc1 complex, whose isoelectric point is shifted toward more alkaline values in the hippocampus of kindled rats. By immunohistochemistry, the Rieske protein is well expressed in the hippocampus, except in the CA1 subfield, an area of selective vulnerability to seizures in humans and animal models. We also noted an asymmetric, selective expression of the Rieske protein in the subgranular neurons of the dorsal dentate gyrus, a region implicated in neurogenesis. CONCLUSIONS: These results indicate that the Rieske protein may play a role in the response of neurons to seizure activity and could give important new insights into the molecular pathogenesis of epilepsy. | 15730530
 |