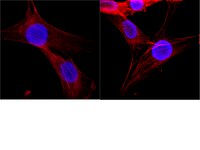
Super-resolution microscopy reveals decondensed chromatin structure at transcription sites.
Wang, Y; Maharana, S; Wang, MD; Shivashankar, GV
Scientific reports
4
4477
2014
Show Abstract
Remodeling of the local chromatin structure is essential for the regulation of gene expression. While a number of biochemical and bioimaging experiments suggest decondensed chromatin structures are associated with transcription, a direct visualization of DNA and transcriptionally active RNA polymerase II (RNA pol II) at super-resolution is still lacking. Here we investigate the structure of chromatin isolated from HeLa cells using binding activatable localization microscopy (BALM). The sample preparation method preserved the structural integrity of chromatin. Interestingly, BALM imaging of the chromatin spreads revealed the presence of decondensed chromatin as gap structures along the spreads. These gaps were enriched with phosphorylated S5 RNA pol II, and were sensitive to the cellular transcriptional state. Taken together, we could visualize the decondensed chromatin regions together with active RNA pol II for the first time using super-resolution microscopy. | | 24667378
 |
Deimination of linker histones links neutrophil extracellular trap release with autoantibodies in systemic autoimmunity.
Dwivedi, N; Neeli, I; Schall, N; Wan, H; Desiderio, DM; Csernok, E; Thompson, PR; Dali, H; Briand, JP; Muller, S; Radic, M
FASEB journal : official publication of the Federation of American Societies for Experimental Biology
28
2840-51
2014
Show Abstract
Autoantibodies to nuclear antigens arise in human autoimmune diseases, but a unifying pathogenetic mechanism remains elusive. Recently we reported that exposure of neutrophils to inflammatory conditions induces the citrullination of core histones by peptidylarginine deiminase 4 (PAD4) and that patients with autoimmune disorders produce autoantibodies that recognize such citrullinated histones. Here we identify histone H1 as an additional substrate of PAD4, localize H1 within neutrophil extracellular traps, and detect autoantibodies to citrullinated H1 in 6% of sera from patients with systemic lupus erythematosus and Sjögren's syndrome. No preference for deiminated H1 was observed in healthy control sera and sera from patients with scleroderma or rheumatoid arthritis. We map binding to the winged helix of H1 and determine that citrulline 53 represents a key determinant of the autoantibody epitope. In addition, we quantitate RNA for H1 histone subtypes in mature human neutrophils and identify citrulline residues by liquid chromatography and tandem mass spectrometry. Our results indicate that deimination of linker histones generates new autoantibody epitopes with enhanced potential for stimulating autoreactive human B cells.-Dwivedi, N., Neeli, I., Schall, N., Wan, H., Desiderio, D. M., Csernok, E., Thompson, P. R., Dali, H., Briand, J.-P., Muller, S., Radic, M. Deimination of linker histones links neutrophil extracellular trap release with autoantibodies in systemic autoimmunity. | | 24671707
 |
The specification and global reprogramming of histone epigenetic marks during gamete formation and early embryo development in C. elegans.
Samson, M; Jow, MM; Wong, CC; Fitzpatrick, C; Aslanian, A; Saucedo, I; Estrada, R; Ito, T; Park, SK; Yates, JR; Chu, DS
PLoS genetics
e1004588
2014
Show Abstract
In addition to the DNA contributed by sperm and oocytes, embryos receive parent-specific epigenetic information that can include histone variants, histone post-translational modifications (PTMs), and DNA methylation. However, a global view of how such marks are erased or retained during gamete formation and reprogrammed after fertilization is lacking. To focus on features conveyed by histones, we conducted a large-scale proteomic identification of histone variants and PTMs in sperm and mixed-stage embryo chromatin from C. elegans, a species that lacks conserved DNA methylation pathways. The fate of these histone marks was then tracked using immunostaining. Proteomic analysis found that sperm harbor ∼2.4 fold lower levels of histone PTMs than embryos and revealed differences in classes of PTMs between sperm and embryos. Sperm chromatin repackaging involves the incorporation of the sperm-specific histone H2A variant HTAS-1, a widespread erasure of histone acetylation, and the retention of histone methylation at sites that mark the transcriptional history of chromatin domains during spermatogenesis. After fertilization, we show HTAS-1 and 6 histone PTM marks distinguish sperm and oocyte chromatin in the new embryo and characterize distinct paternal and maternal histone remodeling events during the oocyte-to-embryo transition. These include the exchange of histone H2A that is marked by ubiquitination, retention of HTAS-1, removal of the H2A variant HTZ-1, and differential reprogramming of histone PTMs. This work identifies novel and conserved features of paternal chromatin that are specified during spermatogenesis and processed in the embryo. Furthermore, our results show that different species, even those with diverged DNA packaging and imprinting strategies, use conserved histone modification and removal mechanisms to reprogram epigenetic information. | Immunofluorescence | 25299455
 |
Nicotine induces chromatin remodelling through decreases in the methyltransferases GLP, G9a, Setdb1 and levels of H3K9me2.
Chase, KA; Sharma, RP
The international journal of neuropsychopharmacology / official scientific journal of the Collegium Internationale Neuropsychopharmacologicum (CINP)
16
1129-38
2013
Show Abstract
Studies examining the epigenetic effects of nicotine are limited, but indicate that nicotine can promote a transcriptionally permissive chromatin environment by increasing acetylation of histone H3 and H4. To further explore nicotine-induced histone modifications, we measured histone methyltransferase (HMT) mRNA expression as well as total and promoter-specific H3K9me2 levels. Following administration of nicotine, HMT mRNA and H3K9me2 levels were examined in mouse primary cortical neuronal culture and cortex extracted from mice injected intraperitoneally, as well as in human lymphocyte culture. Furthermore, Bdnf/BDNF mRNA levels were examined as an epigenetically regulated read-out of gene expression. There was a significant decrease of the HMT GLP, G9a and Setdb1 mRNA expression in the nicotine-treated tissue examined, with significant decreases seen in both total and promoter-specific H3K9me2 levels. Increasing doses of nicotine resulted in significant decreases in Bdnf/BDNF promoter specific H3K9me2 binding, leading to enhanced Bdnf/BDNF transcription. Taken together, our data suggest that nicotine reduces markers of a restrictive epigenomic state, thereby leading to a more permissive epigenomic environment. | | 23067581
 |
Novel alterations in the epigenetic signature of MeCP2-targeted promoters in lymphocytes of Rett syndrome patients.
Lilja, T; Wallenborg, K; Björkman, K; Albåge, M; Eriksson, M; Lagercrantz, H; Rohdin, M; Hermanson, O
Epigenetics
8
246-51
2013
Show Abstract
Rett syndrome (RTT) is a neurodevelopmental disorder with neurological symptoms, such as motor disorders and mental retardation. In most cases, RTT is caused by mutations in the DNA binding protein MeCP2. In mice, MeCP2 gene deletion has been reported to result in genome-wide increased histone acetylation. Transcriptional regulation of neurotrophic factor BDNF and transcription factor DLX5, essential for proper neurogenesis, is further altered in MeCP2-deleted animals. We therefore investigated the chromatin environment of MeCP2 target genes BDNF and DLX5 in lymphocytes from RTT patients and human controls, and analyzed the density of histones H3, H2B and H1, as well as the levels of methylation and acetylation on selected lysines of histone H3. Notably, we found a general increase in the density of histone H3 in RTT patients' lymphocytes compared with controls, and decreased levels of trimethylation of lysine 4 on histone H3 (H3K4me3), a modification associated with transcriptional activation. The levels of acetylation of lysine 9 (H3K9ac) and 27 (H3K27ac) did not show any statistically significant changes when normalized to the decreased histone H3 levels; nevertheless, an average decrease in acetylation was noted. Our results reveal an unexpected alteration of the chromatin state of established MeCP2 target genes in lymphocytes of human subjects with RTT. | | 23348913
 |
High-resolution mapping of h1 linker histone variants in embryonic stem cells.
Cao, K; Lailler, N; Zhang, Y; Kumar, A; Uppal, K; Liu, Z; Lee, EK; Wu, H; Medrzycki, M; Pan, C; Ho, PY; Cooper, GP; Dong, X; Bock, C; Bouhassira, EE; Fan, Y
PLoS genetics
9
e1003417
2013
Show Abstract
H1 linker histones facilitate higher-order chromatin folding and are essential for mammalian development. To achieve high-resolution mapping of H1 variants H1d and H1c in embryonic stem cells (ESCs), we have established a knock-in system and shown that the N-terminally tagged H1 proteins are functionally interchangeable to their endogenous counterparts in vivo. H1d and H1c are depleted from GC- and gene-rich regions and active promoters, inversely correlated with H3K4me3, but positively correlated with H3K9me3 and associated with characteristic sequence features. Surprisingly, both H1d and H1c are significantly enriched at major satellites, which display increased nucleosome spacing compared with bulk chromatin. While also depleted at active promoters and enriched at major satellites, overexpressed H1(0) displays differential binding patterns in specific repetitive sequences compared with H1d and H1c. Depletion of H1c, H1d, and H1e causes pericentric chromocenter clustering and de-repression of major satellites. These results integrate the localization of an understudied type of chromatin proteins, namely the H1 variants, into the epigenome map of mouse ESCs, and we identify significant changes at pericentric heterochromatin upon depletion of this epigenetic mark. | | 23633960
 |
Chromatin context and ncRNA highlight targets of MeCP2 in brain.
Maxwell, SS; Pelka, GJ; Tam, PP; El-Osta, A
RNA biology
10
1741-57
2013
Show Abstract
The discovery that Rett syndrome (RTT) is caused by mutation of the methyl-CpG-binding-protein MeCP2 provided a major breakthrough in understanding the neurodevelopmental disorder and accelerated MeCP2 research. However, gene regulation by MeCP2 is complicated. The current consensus for MeCP2 remains as a classical repressor complex, with major emphasis on its role in methylation-dependent binding and repression. However, recent evidence indicates additional regulatory roles, suggesting non-classical mechanisms in gene activation. This has opened the field of MeCP2 research and suggests that the gene targets may not be the usual suspects, that is, dependent only on DNA methylation. Here we examine how chromatin binding and sequence preference may confer MeCP2 functionality, and connect relevant pathways in an active genome. Finding both genomic and proteomic evidence to indicate MeCP2 spliceosome interaction, we consequently discovered broad MeCP2 enrichment of the transcriptome while our focus toward long non-coding RNA (lncRNA) revealed MeCP2 association with RNCR3. Our data may indicate an as-yet-unappreciated role between lncRNA and MeCP2. We hypothesize that ncRNA may mediate chromatin-remodeling events by interacting with MeCP2, thereby conferring changes in gene expression. We consider that these results may suggest new mechanisms of gene regulation conferred by MeCP2 and its interactions upon chromatin structure and gene function. | | 24270455
 |
Prostate cancer stem cell-targeted efficacy of a new-generation taxoid, SBT-1214 and novel polyenolic zinc-binding curcuminoid, CMC2.24.
Botchkina, GI; Zuniga, ES; Rowehl, RH; Park, R; Bhalla, R; Bialkowska, AB; Johnson, F; Golub, LM; Zhang, Y; Ojima, I; Shroyer, KR
PloS one
8
e69884
2013
Show Abstract
Prostate cancer is the second leading cause of cancer death among men. Multiple evidence suggests that a population of tumor-initiating, or cancer stem cells (CSCs) is responsible for cancer development and exceptional drug resistance, representing a highly important therapeutic target. The present study evaluated CSC-specific alterations induced by new-generation taxoid SBT-1214 and a novel polyenolic zinc-binding curcuminoid, CMC2.24, in prostate CSCs.The CD133(high)/CD44(high) phenotype was isolated from spontaneously immortalized patient-derived PPT2 cells and highly metastatic PC3MM2 cells. Weekly treatment of the NOD/SCID mice bearing PPT2- and PC3MM3-induced tumors with the SBT-1214 led to dramatic suppression of tumor growth. Four of six PPT2 and 3 of 6 PC3MM2 tumors have shown the absence of viable cells in residual tumors. In vitro, SBT-1214 (100 nM-1 µM; for 72 hr) induced about 60% cell death in CD133(high)/CD44(+/high) cells cultured on collagen I in stem cell medium (in contrast, the same doses of paclitaxel increased proliferation of these cells). The cytotoxic effects were increased when SBT-1214 was combined with the CMC2.24. A stem cell-specific PCR array assay revealed that this drug combination mediated massive inhibition of multiple constitutively up-regulated stem cell-related genes, including key pluripotency transcription factors. Importantly, this drug combination induced expression of p21 and p53, which were absent in CD133(high)/CD44(high) cells. Viable cells that survived this treatment regimen were no longer able to induce secondary spheroids, exhibited significant morphological abnormalities and died in 2-5 days.We report here that the SBT-1214 alone, or in combination with CMC2.24, possesses significant activity against prostate CD133(high)/CD44(+/high) tumor-initiating cells. This drug combination efficiently inhibits expression of the majority of stem cell-related genes and pluripotency transcription factors. In addition, it induces a previously absent expression of p21 and p53 ("gene wake-up"), which can potentially reverse drug resistance by increasing sensitivity to anti-cancer drugs. | | 24086245
 |
Histone h1 depletion impairs embryonic stem cell differentiation.
Zhang, Y; Cooke, M; Panjwani, S; Cao, K; Krauth, B; Ho, PY; Medrzycki, M; Berhe, DT; Pan, C; McDevitt, TC; Fan, Y
PLoS genetics
8
e1002691
2012
Show Abstract
Pluripotent embryonic stem cells (ESCs) are known to possess a relatively open chromatin structure; yet, despite efforts to characterize the chromatin signatures of ESCs, the role of chromatin compaction in stem cell fate and function remains elusive. Linker histone H1 is important for higher-order chromatin folding and is essential for mammalian embryogenesis. To investigate the role of H1 and chromatin compaction in stem cell pluripotency and differentiation, we examine the differentiation of embryonic stem cells that are depleted of multiple H1 subtypes. H1c/H1d/H1e triple null ESCs are more resistant to spontaneous differentiation in adherent monolayer culture upon removal of leukemia inhibitory factor. Similarly, the majority of the triple-H1 null embryoid bodies (EBs) lack morphological structures representing the three germ layers and retain gene expression signatures characteristic of undifferentiated ESCs. Furthermore, upon neural differentiation of EBs, triple-H1 null cell cultures are deficient in neurite outgrowth and lack efficient activation of neural markers. Finally, we discover that triple-H1 null embryos and EBs fail to fully repress the expression of the pluripotency genes in comparison with wild-type controls and that H1 depletion impairs DNA methylation and changes of histone marks at promoter regions necessary for efficiently silencing pluripotency gene Oct4 during stem cell differentiation and embryogenesis. In summary, we demonstrate that H1 plays a critical role in pluripotent stem cell differentiation, and our results suggest that H1 and chromatin compaction may mediate pluripotent stem cell differentiation through epigenetic repression of the pluripotency genes. | | 22589736
 |
Vitamin D receptor activators upregulate and rescue podocalyxin expression in high glucose-treated human podocytes.
Verouti, SN; Tsilibary, EC; Fragopoulou, E; Iatrou, C; Demopoulos, CA; Charonis, AS; Charonis, SA; Drossopoulou, GI
Nephron Exp Nephrol
122
36-50
2012
Show Abstract
Vitamin D is beneficial in human and experimental chronic kidney disease, the leading cause of which is diabetic nephropathy. Vitamin D through its receptor, VDR, provides renal protection in diabetic nephropathy, but limited data exist about its effect on podocytes. Renal podocytes form the main filtration barrier possessing a unique phenotype maintained by proteins including podocalyxin and nephrin, the expression of which is suppressed in pathological conditions.We used immortalized human podocytes (human glomerular epithelial cells, HGEC) to assess podocalyxin and nephrin expression after treatment with 1,25-dihydroxyvitamin D3 (calcitriol) and its analogue paricalcitol. The involvement of VDR was investigated by silencing with hVDR-siRNA and ChIP analysis.HGEC exhibit high glucose-mediated downregulation of podocalyxin and nephrin, loss of which has been linked with loss of the permselective renal barrier and proteinuria. Calcitriol and paricalcitol reversed high glucose-induced decrease of nephrin and significantly enhanced podocalyxin expression in podocytes cultured in high glucose. HGEC express VDR and retinoid X receptor (RXR). In the presence of calcitriol and paricalcitol, VDR expression was upregulated and VDR colocalized with RXR in the nucleus. VDR knockdown abolished the protective action of calcitriol and paricalcitol on podocalyxin expression indicating that podocalyxin activation of expression is partly mediated by VDR. Furthermore, VDR specifically regulates podocalyxin expression by bounding to a site upstream of the podocalyxin promoter.Vitamin D analogues maintain and, furthermore, re-activate the expression of specialized components of podocytes including podocalyxin, hence they provide protection against loss of the permselective renal barrier, with molecular mechanisms elucidated herein. | Fluorescence Activated Cell Sorting (FACS) | 23548800
 |





















-Cinchonine[802506_(+)-Cinchonine-ALL].jpg)