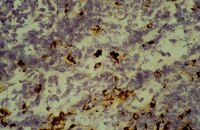
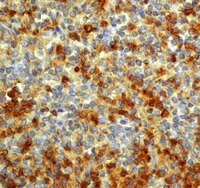
Hypertonic saline alleviates cerebral edema by inhibiting microglia-derived TNF-α and IL-1β-induced Na-K-Cl Cotransporter up-regulation.
Huang, LQ; Zhu, GF; Deng, YY; Jiang, WQ; Fang, M; Chen, CB; Cao, W; Wen, MY; Han, YL; Zeng, HK
Journal of neuroinflammation
11
102
2014
Show Abstract
Hypertonic saline (HS) has been successfully used clinically for treatment of various forms of cerebral edema. Up-regulated expression of Na-K-Cl Cotransporter 1 (NKCC1) and inflammatory mediators such as tumor necrosis factor alpha (TNF-α) and interleukin-1 beta (IL-1β) has been demonstrated to be closely associated with the pathogenesis of cerebral edema resulting from a variety of brain injuries. This study aimed to explore if alleviation of cerebral edema by 10% HS might be effected through down-regulation of inflammatory mediator expression in the microglia, and thus result in decreased NKCC1 expression in astrocytes in the cerebral cortex bordering the ischemic core.The Sprague-Dawley (SD) rats that underwent right-sided middle cerebral artery occlusion (MCAO) were used for assessment of NKCC1, TNF-α and IL-1β expression using Western blotting, double immunofluorescence and real time RT-PCR, and the model also was used for evaluation of brain water content (BWC) and infarct size. SB203580 and SP600125, specific inhibitors of the p38 and JNK signaling pathways, were used to treat primary microglia cultures to determine whether the two signaling pathways were required for the inhibition of HS on microglia expressing and secreting TNF-α and IL-1β using Western blotting, double immunofluorescence and enzyme-linked immunosorbent assay (ELISA). The effect of TNF-α and IL-1β on NKCC1 expression in primary astrocyte cultures was determined. In addition, the direct inhibitory effect of HS on NKCC1 expression in primary astrocytes was also investigated by Western blotting, double immunofluorescence and real time RT-PCR.BWC and infarct size decreased significantly after 10% HS treatment. TNF-α and IL-1β immunoexpression in microglia was noticeably decreased. Concomitantly, NKCC1 expression in astrocytes was down-regulated. TNF-α and IL-1β released from the primary microglia subjected to hypoxic exposure and treatment with 100 mM HS were decreased. NKCC1 expression in primary astrocytes was concurrently and progressively down-regulated with decreasing concentration of exogenous TNF-α and IL-1β. Additionally, 100 mM HS directly inhibited NKCC1 up-regulation in astrocytes under hypoxic condition.The results suggest that 10% HS alleviates cerebral edema through inhibition of the NKCC1 Cotransporter, which is mediated by attenuation of TNF-α and IL-1β stimulation on NKCC1. | 24916922
 |
Impact of vegf on astrocytes: analysis of gap junctional intercellular communication, proliferation, and motility.
Ricarda Wuestefeld,Jingchen Chen,Karl Meller,Beate Brand-Saberi,Carsten Theiss
Glia
60
2012
Show Abstract
The purpose of the present study was to investigate the effects of vascular endothelial growth factor (VEGF) on gap junctional intercellular communication (GJIC), cell proliferation, and cell dynamics in primary astrocytes. VEGF is known as a dimeric polypeptide that potentially binds to two receptors, VEGFR-1 and VEGFR-2, however many effects are mediated by VEGFR-2, for example, actin polymerization, forced cell migration, angiogenesis, and cell proliferation. Recently it has been shown that in case of hypoxia, ischemia or injury VEGF is upregulated to stimulate angiogenesis and cell proliferation. Besides this, VEGF reveals a potent therapeutical target for averting tumor vascularization, emerging in bevacizumab, the first humanized anti-VEGF-A antibody for treating recurrent Glioblastoma multiforme. To expand our knowledge about VEGF effects in glial cells, we cultivated rat astrocytes in medium containing VEGF for 1 and 2 days. To investigate the effects of VEGF on GJIC, we microinjected neurobiotin into a single cell and monitored dye-spreading into adjacent cells. These experiments showed that VEGF significantly enhances astrocytic GJIC compared with controls. Cell proliferation measured by BrdU-labeling also revealed a significant increase of astrocytic mitose rates subsequent to 1 day of VEGF exposure, whereas longer VEGF treatment for 2 days did not have additive effects. To study cell-dynamics of astrocytes subsequent to VEGF treatment, we additionally transfected astrocytes with LifeAct-RFP. Live-cell imaging and quantitative analysis of these cells with aid of confocal laser scanning microscopy revealed higher process movement of VEGF-treated astrocytes. In conclusion, VEGF strongly affects cell proliferation, GJIC, and motility in astrocytes. | 22431192
 |
Stainless steel ions stimulate increased thrombospondin-1-dependent TGF-beta activation by vascular smooth muscle cells: implications for in-stent restenosis.
Pallero, MA; Talbert Roden, M; Chen, YF; Anderson, PG; Lemons, J; Brott, BC; Murphy-Ullrich, JE
Journal of vascular research
47
309-22
2010
Show Abstract
Despite advances in stent design, in-stent restenosis (ISR) remains a significant clinical problem. All implant metals exhibit corrosion, which results in release of metal ions. Stainless steel (SS), a metal alloy widely used in stents, releases ions to the vessel wall and induces reactive oxygen species, inflammation and fibroproliferative responses. The molecular mechanisms are unknown. TGF-beta is known to be involved in the fibroproliferative responses of vascular smooth muscle cells (VSMCs) in restenosis, and TGF-beta antagonists attenuate ISR. We hypothesized that SS ions induce the latent TGF-beta activator, thrombospondin-1 (TSP1), through altered oxidative signaling to stimulate increased TGF-beta activation and VSMC phenotype change.VSMCs were treated with SS metal ion cocktails, and morphology, TSP1, extracellular matrix production, desmin and TGF-beta activity were assessed by immunoblotting.SS ions stimulate the synthetic phenotype, increased TGF-beta activity, TSP1, increased extracellular matrix and downregulation of desmin in VSMCs. Furthermore, SS ions increase hydrogen peroxide and decrease cGMP-dependent protein kinase (PKG) signaling, a known repressor of TSP1 transcription. Catalase blocks SS ion attenuation of PKG signaling and increased TSP1 expression.These data suggest that ions from stent alloy corrosion contribute to ISR through stimulation of TSP1-dependent TGF-beta activation. Full Text Article | 20016205
 |
Erythropoietin receptor expression is concordant with erythropoietin but not with common beta chain expression in the rat brain throughout the life span.
Pascal E Sanchez,Fabrice P Navarro,Raafat P Fares,Jérémie Nadam,Béatrice Georges,Colette Moulin,Marion Le Cavorsin,Chantal Bonnet,Philippe Ryvlin,Amor Belmeguenai,Jacques Bodennec,Anne Morales,Laurent Bezin
The Journal of comparative neurology
514
2009
Show Abstract
Brain effects of erythropoietin (Epo) are proposed to involve a heteromeric receptor comprising the classical Epo receptor (Epo-R) and the common beta chain (betac). However, data documenting the pattern of betac gene expression in the healthy brain, in comparison with that of the Epo-R gene, are still lacking. The present study is the first to investigate at the same time betac, Epo-R, and Epo gene expression within different rat brain areas throughout the life span, from neonatal to elderly stages, using quantitative RT-PCR for transcripts. Corresponding proteins were localized by using immunohistochemistry. We demonstrate that the betac transcript level does not correlate with that of Epo-R or Epo, whereas the Epo-R transcript level strongly correlates with that of Epo throughout the life span in all brain structures analyzed. Both Epo and Epo-R were detected primarily in neurons. In the hippocampus, the greatest Epo-R mRNA levels were measured during the early postnatal period and in middle-aged rats, associated with an intense neuronal immunolabeling. Conversely, betac protein was barely detectable in the brain at all ages, even in neurons expressing high levels of Epo-R. Finally, betac transcript could not be detected in PC12 cells, even after nerve growth factor-induced neuritogenesis, which is a condition that dramatically enhances Epo-R transcript level. Altogether, our data suggest that most neurons are likely to express high levels of Epo-R but low, if not null, levels of betac. Given that Epo protects extended populations of neurons after injury, a yet-to-be-identified receptor heterocomplex including Epo-R may exist in the large population of brain neurons that does not express betac. | 19330822
 |
A protocol for phenotypic detection and characterization of vascular cells of different origins in a lung neovascularization model in rodents.
Jones, RC; Capen, DE; Cohen, KS; Munn, LL; Jain, RK; Duda, DG
Nature protocols
3
388-97
2008
Show Abstract
The goal of many current studies of neovascularization is to define the phenotype of vascular cell populations of different origins and to determine how such cells promote assembly of vascular channel. Here, we describe a protocol to immunophenotype vascular cells by high-resolution imaging and by fluorescence-activated flow cytometry in an in vivo rodent model of pulmonary microvascular remodeling. Analysis of cells by this combined approach will characterize their phenotype, quantify their number and identify their role in the assembly of vascular channels. | 18323810
 |
Neuroprotective effects of erythropoietin in the rat hippocampus after pilocarpine-induced status epilepticus.
Jérémie Nadam, Fabrice Navarro, Pascal Sanchez, Colette Moulin, Béatrice Georges, Aël Laglaine, Jean-Marc Pequignot, Anne Morales, Philippe Ryvlin, Laurent Bezin
Neurobiology of disease
25
412-26
2007
Show Abstract
Neuroprotective functions of erythropoietin (Epo) are thought to involve a heteroreceptor composed of both Epo receptor (Epo-R) and common beta chain (betac). Here, we measured the response of hippocampal Epo system components (Epo, Epo-R and betac) during neurodegenerative processes following pilocarpine-induced status epilepticus (SE), and examined whether recombinant human Epo (rHuEpo) could support neuronal survival. We evidence that Epo is induced in astroglia following SE, in particular within areas displaying delayed neuronal death. In addition, we demonstrate for the first time that rHuEpo reduces considerably hippocampal neurodegeneration following SE. rHuEpo may thus supplement astroglial induction of Epo to promote enhanced hippocampal neuronal survival following SE. We also show that Epo-R is expressed by neurons and astrocytes mainly, while betac is barely detectable in basal conditions and induced in reactive microglia exclusively following SE. Altogether, our results suggest that Epo/rHuEpo exerts neuroprotection, through Epo-R signaling and independently of betac, and, therefore, may be anti-epileptogenic. | 17166730
 |
Immunohistochemical detection of T-cell subsets and other leukocytes in paraffin-embedded rat and mouse tissues with monoclonal antibodies.
Whiteland, J L, et al.
J. Histochem. Cytochem., 43: 313-20 (1995)
1995
Show Abstract
We describe a method for immunohistochemical localization of T-cells, CD4+ T-cells, CD8+ T-cells, B-cells, activated lymphocytes, major histocompatibility complex (MHC) class II antigens, macrophages, dendritic cells, and granulocytes in rat and mouse tissue fixed in periodate-lysine-paraformaldehyde (PLP) and embedded in paraffin. Rat and mouse spleen and eyes were fixed in PLP for 18-24 hr, rapidly dehydrated, infiltrated under vacuum with paraffin at 54 degrees C, sectioned, and stained with appropriate monoclonal antibodies (MAbs). Sections of PLP-fixed, paraffin-embedded spleen were compared with acetone-fixed frozen spleen sections with respect to morphology and staining quality. Nine of 10 MAbs to rat antigens and eight of nine MAbs to mouse antigens stained paraffin sections equally or more intensely than frozen sections. The two MAbs that showed weaker staining still gave good staining on paraffin sections. Paraffin-embedded rat and mouse eyes were easier to section serially than frozen eyes, showed superior morphology, and individually stained cells were readily identified. Therefore, a combination of PLP fixation and low-temperature paraffin embedding permits detection of the major types of immune cell in rat and mouse tissues while maintaining good morphology, particularly in diseased, damaged, or delicate tissues. | 7868861
 |
Differential immunochemical markers reveal the normal distribution of brain macrophages and microglia in the developing rat brain
Milligan C.E. et al.
J Comparative Neurol., 314:125-135 (1991)
1991
| 1797868
 |
Macrophage heterogeneity in the rat as delineated by two monoclonal antibodies MRC OX-41 and MRC OX-42, the latter recognizing complement receptor type 3
Robinson A.P. et al.
Immunol., 57:239-247 (1986)
1986
| 3512425
 |