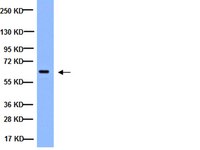
Bryostatin activates HIV-1 latent expression in human astrocytes through a PKC and NF-ĸB-dependent mechanism.
Díaz, L; Martínez-Bonet, M; Sánchez, J; Fernández-Pineda, A; Jiménez, JL; Muñoz, E; Moreno, S; Álvarez, S; Muñoz-Fernández, MÁ
Scientific reports
5
12442
2015
Show Abstract
Multiple studies have shown that HIV-1 patients may develop virus reservoirs that impede eradication; these reservoirs include the central nervous system (CNS). Despite an undetectable viral load in patients treated with potent antiretrovirals, current therapy is unable to purge the virus from these latent reservoirs. To broaden the inhibitory range and effectiveness of current antiretrovirals, the potential of bryostatin was investigated as a latent HIV-1 activator. We used primary astrocytes, NHA cells, and astrocytoma cells U-87. Infected cells with HIV-1(NL4.3) were treated with bryostatin alone or in combination with different inhibitors. HIV-1 production was quantified by using ELISA. Transcriptional activity was measured using luciferase reporter gene assays by using lipofectin. We performed cotransfection experiments of the LTR promoter with the active NF-κB member p65/relA. To confirm the NF-κB role, Western blot and confocal microscopy were performed. Bryostatin reactivates latent viral infection in the NHA and U87 cells via activation of protein kinase C (PKC)-alpha and -delta, because the PKC inhibitors rottlerin and GF109203X abrogated the bryostatin effect. No alteration in cell proliferation was found. Moreover, bryostatin strongly stimulated LTR transcription by activating the transcription factor NF-κB. Bryostatin could be a beneficial adjunct to the treatment of HIV-1 brain infection. | | | 26199173
 |
PTPIP51—A New RelA-tionship with the NFκB Signaling Pathway.
Brobeil, A; Kämmerer, F; Tag, C; Steger, K; Gattenlöhner, S; Wimmer, M
Biomolecules
5
485-504
2015
Show Abstract
The present study shows a new connection of protein tyrosine phosphatase interacting protein 51 (PTPIP51) to the nuclear factor κB (NFκB) signalling pathway. PTPIP51 mRNA and protein expression is regulated by RelA. If bound to the PTPIP51 promoter, RelA repress the mRNA and protein expression of PTPIP51. The parallel treatment with pyrrolidine dithiocarbamate (PDTC) reversed the suppression of PTPIP51 protein expression induced by TNFα. Using the intensity correlation analysis PTPIP51 verified a co-localization with RelA, which is also regulated by TNFα administration. Moreover, the direct interaction of PTPIP51 and RelA was established using the DuoLink proximity ligation assay. IκBα, the known inhibitor of RelA, also interacted with PTPIP51. This hints to the fact that in un-stimulated conditions PTPIP51 forms a complex with RelA and IκBα. The PTPIP51/RelA/IκBα complex is modulated by TNFα. Interestingly, the impact on the mitogen activated protein kinase pathway was negligible except in highest TNFα concentration. Here, PTPIP51 and Raf-1 interactions were slightly repressed. The newly established relationship of PTPIP51 and the NFκB signaling pathway provides the basis for a possible therapeutic impact. | | | 25893721
 |
An investigation of the specificity of research antibodies against NF-κB-subunit p65.
Slotta, C; Müller, J; Tran, L; Hauser, S; Widera, D; Kaltschmidt, B; Kaltschmidt, C
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society
62
157-61
2014
Show Abstract
0 | | | 24255050
 |
An Immunomodulatory Protein (Ling Zhi-8) from a Ganoderma lucidum Induced Acceleration of Wound Healing in Rat Liver Tissues after Monopolar Electrosurgery.
Lin, HJ; Chang, YS; Lin, LH; Haung, CF; Wu, CY; Ou, KL
Evidence-based complementary and alternative medicine : eCAM
2014
916531
2014
Show Abstract
The purpose of this study was to investigate the effect of an immunomodulatory protein (Ling Zhi-8, LZ-8) on wound healing in rat liver tissues after monopolar electrosurgery. Animals were sacrificed for evaluations at 0, 3, 7, and 28 days postoperatively. It was found that the wound with the LZ-8 treatment significantly increases wound healing. Western blot analysis clearly indicated that the expression of NF-κB was decreased at 3, 7, and 28 days when liver tissues were treated with LZ-8. Moreover, caspase-3 activity of the liver tissue also significantly decreases at 7 and 28 days, respectively. DAPI staining and TUNEL assays revealed that only a minimal dispersion of NF-κB was found on the liver tissue treated with LZ-8 at day 7 as compared with day 3 and tissues without LZ-8 treatment. Similarly, apoptosis was decreased on liver tissues treated with LZ-8 at 7 days when compared to the control (monopolar electrosurgery) tissues. Therefore, the analytical results demonstrated that LZ-8 induced acceleration of wound healing in rat liver tissues after monopolar electrosurgery. | | | 24883073
 |
Nuclear factor kappa-B signaling is integral to ocular neovascularization in ischemia-independent microenvironment.
DeNiro, M; Al-Mohanna, FA
PloS one
9
e101602
2014
Show Abstract
Retinal ischemia promotes the upregulation of VEGF expression and accounts for most pathological features of retinal neovascularization (NV). Paradoxically, VEGF remains the pivotal stimulator of ocular NV, despite the absence of ischemia. Therefore, the central question arises as to how the various molecular mechanisms interplay in ischemia-independent NV. It's been suggested that NFκB plays a crucial role in the pathogenesis of diabetic vasculopathies. Here, we dissected the molecular mechanism of ocular NV in the rho/VEGF transgenic mouse model, which develops subretinal NV in ischemia-independent microenvironment. Furthermore, we examined whether intravitreal administration of YC-1, a HIF-1 inhibitor, can modulate the activation of NFκB and its downstream angiogenic signaling in the mouse retina. We demonstrated that YC-1 inhibited retinal NFκB/p65 DNA binding activity and downregulated NFκB/p65, FAK, α5β1, EPO, ET-1, and MMP-9 expression at the message and the protein levels. In addition, YC-1 significantly inhibited subretinal NV by reducing the number of neovascular lesions, the area of each lesion and the total area of NV per retina. We further investigated the influence of VEGF signaling pathway on HIF-1α transcriptional activity to substantiate that this mouse model develops subretinal NV in an ischemia-independent microenvironment. Our data demonstrated that VEGF overexpression didn't have any impact on HIF-1α transcriptional activity, whereas treatment with YC-1 significantly inhibited endogenous HIF-1 activity. Our study suggests that retinal NFκB transcriptional activity is pivotal to ischemia-independent mechanisms, which lead to the local activation of angiogenic cascades. Our data also indicate that the nexus between VEGF and NFκB is implicated in triggering the angiogenic cascade that promotes retinal NV. Hence, targeting the VEGF/NFκB axis may act in a negative feedback loop to suppress ocular NV. This study suggests that inhibition of NFκB activation may be a means of turning off a "master switch" responsible for initiating and perpetuating these ocular pathologies. | | | 25050547
 |
Theta-burst stimulation of hippocampal slices induces network-level calcium oscillations and activates analogous gene transcription to spatial learning.
Sheridan, GK; Moeendarbary, E; Pickering, M; O'Connor, JJ; Murphy, KJ
PloS one
9
e100546
2014
Show Abstract
Over four decades ago, it was discovered that high-frequency stimulation of the dentate gyrus induces long-term potentiation (LTP) of synaptic transmission. LTP is believed to underlie how we process and code external stimuli before converting it to salient information that we store as 'memories'. It has been shown that rats performing spatial learning tasks display theta-frequency (3-12 Hz) hippocampal neural activity. Moreover, administering theta-burst stimulation (TBS) to hippocampal slices can induce LTP. TBS triggers a sustained rise in intracellular calcium [Ca2+]i in neurons leading to new protein synthesis important for LTP maintenance. In this study, we measured TBS-induced [Ca2+]i oscillations in thousands of cells at increasing distances from the source of stimulation. Following TBS, a calcium wave propagates radially with an average speed of 5.2 µm/s and triggers multiple and regular [Ca2+]i oscillations in the hippocampus. Interestingly, the number and frequency of [Ca2+]i fluctuations post-TBS increased with respect to distance from the electrode. During the post-tetanic phase, 18% of cells exhibited 3 peaks in [Ca2+]i with a frequency of 17 mHz, whereas 2.3% of cells distributed further from the electrode displayed 8 [Ca2+]i oscillations at 33 mHz. We suggest that these observed [Ca2+]i oscillations could lead to activation of transcription factors involved in synaptic plasticity. In particular, the transcription factor, NF-κB, has been implicated in memory formation and is up-regulated after LTP induction. We measured increased activation of NF-κB 30 min post-TBS in CA1 pyramidal cells and also observed similar temporal up-regulation of NF-κB levels in CA1 neurons following water maze training in rats. Therefore, TBS of hippocampal slice cultures in vitro can mimic the cell type-specific up-regulations in activated NF-κB following spatial learning in vivo. This indicates that TBS may induce similar transcriptional changes to spatial learning and that TBS-triggered [Ca2+]i oscillations could activate memory-associated gene expression. | | | 24950243
 |
An adeno-associated virus-based intracellular sensor of pathological nuclear factor-κB activation for disease-inducible gene transfer.
Chtarto, A; Bockstael, O; Gebara, E; Vermoesen, K; Melas, C; Pythoud, C; Levivier, M; De Witte, O; Luthi-Carter, R; Clinkers, R; Tenenbaum, L
PloS one
8
e53156
2013
Show Abstract
Stimulation of resident cells by NF-κB activating cytokines is a central element of inflammatory and degenerative disorders of the central nervous system (CNS). This disease-mediated NF-κB activation could be used to drive transgene expression selectively in affected cells, using adeno-associated virus (AAV)-mediated gene transfer. We have constructed a series of AAV vectors expressing GFP under the control of different promoters including NF-κB -responsive elements. As an initial screen, the vectors were tested in vitro in HEK-293T cells treated with TNF-α. The best profile of GFP induction was obtained with a promoter containing two blocks of four NF-κB -responsive sequences from the human JCV neurotropic polyoma virus promoter, fused to a new tight minimal CMV promoter, optimally distant from each other. A therapeutical gene, glial cell line-derived neurotrophic factor (GDNF) cDNA under the control of serotype 1-encapsidated NF-κB -responsive AAV vector (AAV-NF) was protective in senescent cultures of mouse cortical neurons. AAV-NF was then evaluated in vivo in the kainic acid (KA)-induced status epilepticus rat model for temporal lobe epilepsy, a major neurological disorder with a central pathophysiological role for NF-κB activation. We demonstrate that AAV-NF, injected in the hippocampus, responded to disease induction by mediating GFP expression, preferentially in CA1 and CA3 neurons and astrocytes, specifically in regions where inflammatory markers were also induced. Altogether, these data demonstrate the feasibility to use disease-activated transcription factor-responsive elements in order to drive transgene expression specifically in affected cells in inflammatory CNS disorders using AAV-mediated gene transfer. | | | 23301037
 |
The nexus between VEGF and NFκB orchestrates a hypoxia-independent neovasculogenesis.
DeNiro, M; Al-Mohanna, FH; Alsmadi, O; Al-Mohanna, FA
PloS one
8
e59021
2013
Show Abstract
Nuclear Factor-Kappa B [NFκB] activation triggers the elevation of various pro-angiogenic factors that contribute to the development and progression of diabetic vasculopathies. It has been demonstrated that vascular endothelial growth factor [VEGF] activates NFκB signaling pathway. Under the ischemic microenvironments, hypoxia-inducible factor-1 [HIF-1] upregulates the expression of several proangiogenic mediators, which play crucial roles in ocular pathologies. Whereas YC-1, a soluble guanylyl cyclase [sGC] agonist, inhibits HIF-1 and NFκB signaling pathways in various cell and animal models. Throughout this investigation, we examined the molecular link between VEGF and NFκB under a hypoxia-independent microenvironment in human retinal microvascular endothelial cells [hRMVECs]. Our data indicate that VEGF promoted retinal neovasculogenesis via NFκB activation, enhancement of its DNA-binding activity, and upregulating NFκB/p65, SDF-1, CXCR4, FAK, αVβ3, α5β1, EPO, ET-1, and MMP-9 expression. Conversely, YC-1 impaired the activation of NFκB and its downstream signaling pathways, via attenuating IκB kinase phosphorylation, degradation and activation, and thus suppressing p65 phosphorylation, nuclear translocation, and inhibiting NFκB-DNA binding activity. We report for the first time that the nexus between VEGF and NFκB is implicated in coordinating a scheme that upregulates several pro-angiogenic molecules, which promotes retinal neovasculogenesis. Our data may suggest the potential use of YC-1 to attenuate the deleterious effects that are associated with hypoxia/ischemia-independent retinal vasculopathies. | | | 23533599
 |
Novel signaling mechanisms of intracellular angiotensin II-induced NHE3 expression and activation in mouse proximal tubule cells.
Li, XC; Hopfer, U; Zhuo, JL
American journal of physiology. Renal physiology
303
F1617-28
2012
Show Abstract
Expression of a cytosolic cyan fluorescent fusion protein of angiotensin II (ECFP/ANG II) in proximal tubules increases blood pressure in rodents. To determine cellular signaling pathways responsible for this response, we expressed ECFP/ANG II in transport-competent mouse proximal convoluted tubule cells (mPCT) from wild-type (WT) and type 1a ANG II receptor-deficient (AT(1a)-KO) mice and measured its effects on intracellular ANG II levels, surrogates of Na/H exchanger 3 (NHE3)-dependent Na(+) absorption, as well as MAP kinases and NF-κB signaling. In WT mPCT cells, ECFP/ANG II expression doubled ANG II levels, increased NHE3 expression and membrane phospho-NHE3 proteins threefold and intracellular Na(+) concentration by 65%. These responses were associated with threefold increases in phospho-ERK 1/2 and phospho-p38 MAPK, fivefold increases in p65 subunit of NF-κB, and threefold increases in phospho-IKKα/β (Ser 176/180) proteins. These signaling responses to ECFP/ANG II were inhibited by losartan (AT(1) blocker), PD123319 (AT(2) blocker), U0126 (MEK1/MEK2 inhibitor), and RO 106-9920 (NF-κB inhibitor). In mPCT cells of AT(1a)-KO mice, ECFP/ANG II also increased the levels of NHE3, p-ERK1/2, and p65 proteins above their controls, but considerably less so than in WT cells. In WT mice, selective expression of ECFP/ANG II in vivo in proximal tubules significantly increased blood pressure and indices of sodium reabsorption, in particular levels of phosphorylated NHE3 protein in the membrane fraction and proton gradient-stimulated (22)Na(+) uptake by proximal tubules. We conclude that intracellular ANG II may induce NHE3 expression and activation in mPCTs via AT(1a)- and AT(2) receptor-mediated activation of MAP kinases ERK 1/2 and NF-κB signaling pathways. | | | 23034941
 |
Accumulation of argpyrimidine, a methylglyoxal-derived advanced glycation end product, increases apoptosis of lens epithelial cells both in vitro and in vivo.
Kim, J; Kim, OS; Kim, CS; Sohn, E; Jo, K; Kim, JS
Experimental & molecular medicine
44
167-75
2012
Show Abstract
The formation of advanced glycation end products (AGEs) has been considered to be a potential causative factor of injury to lens epithelial cells (LECs). Damage of LECs is believed to contribute to cataract formation. The purpose of this study was to investigate the cytotoxic effect of AGEs on LECs both in vitro and in vivo. We examined the accumulation of argpyrimidine, a methylglyoxal-derived AGE, and the expression of apoptosis-related molecules including nuclear factor- kappaB (NF-κB), Bax, and Bcl-2 in the human LEC line HLE-B3 and in cataractous lenses of Zucker diabetic fatty (ZDF) rats, an animal model of type 2 diabetes. In cataractous lenses from twenty-oneweek- old ZDF rats, LEC apoptosis was markedly increased, and the accumulation of argpyrimidine as well as subsequent activation of NF-κB in LECs were significantly enhanced. The ratio of Bax to Bcl-2 protein levels was also increased. In addition, the accumulation of argpyrimidine triggered apoptosis in methylglyoxal- treated HLE-B3 cells. However, the presence of pyridoxamine (an AGEs inhibitor) and pyrrolidine dithiocarbamate (a NF-κB inhibitor) prevented apoptosis in HLE-B3 cells through the inhibition of argpyrimidine formation and the blockage of NF-κB nuclear translocalization, respectively. These results suggest that the cellular accumulation of argpyrimidine in LECs is NF-κB-dependent and pro-apoptotic. | | | 22139526
 |