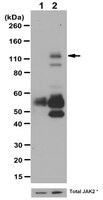
15-Lipoxygenase-1-enhanced Src-Janus kinase 2-signal transducer and activator of transcription 3 stimulation and monocyte chemoattractant protein-1 expression require redox-sensitive activation of epidermal growth factor receptor in vascular wall remodeling.
Singh, NK; Wang, D; Kundumani-Sridharan, V; Van Quyen, D; Niu, J; Rao, GN
The Journal of biological chemistry
286
22478-88
2011
Show Abstract
To understand the mechanisms by which 15(S)-hydroxyeicosatetraenoic acid (15(S)-HETE) activates signal transducer and activator of transcription 3 (STAT3), we studied the role of epidermal growth factor receptor (EGFR). 15(S)-HETE stimulated tyrosine phosphorylation of EGFR in a time-dependent manner in vascular smooth muscle cells (VSMCs). Interference with EGFR activation blocked 15(S)-HETE-induced Src and STAT3 tyrosine phosphorylation, monocyte chemoattractant protein-1 (MCP-1) expression and VSMC migration. 15(S)-HETE also induced tyrosine phosphorylation of Janus kinase 2 (Jak2) in VSMCs, and its inhibition substantially reduced STAT3 phosphorylation, MCP-1 expression, and VSMC migration. In addition, Src formed a complex with EGFR and Jak2, and its inhibition completely blocked Jak2 and STAT3 phosphorylation, MCP-1 expression, and VSMC migration. 15(S)-HETE induced the production of H(2)O(2) via an NADPH oxidase-dependent manner and its scavengers, N-acetyl cysteine (NAC) and catalase suppressed 15(S)-HETE-stimulated EGFR, Src, Jak2, and STAT3 phosphorylation and MCP-1 expression. Balloon injury (BI) induced EGFR, Src, Jak2, and STAT3 phosphorylation, and inhibition of these signaling molecules attenuated BI-induced MCP-1 expression and smooth muscle cell migration from the medial to the luminal surface resulting in reduced neointima formation. In addition, inhibition of EGFR blocked BI-induced Src, Jak2, and STAT3 phosphorylation. Similarly, interference with Src activation suppressed BI-induced Jak2 and STAT3 phosphorylation. Furthermore, adenovirus-mediated expression of dnJak2 also blocked BI-induced STAT3 phosphorylation. Consistent with the effects of 15(S)-HETE on the activation of EGFR-Src-Jak2-STAT3 signaling in VSMCs in vitro, adenovirus-mediated expression of 15-lipoxygenase 1 (15-Lox1) enhanced BI-induced EGFR, Src, Jak2, and STAT3 phosphorylation leading to enhanced MCP-1 expression in vivo. Blockade of Src or Jak2 suppressed BI-induced 15-Lox1-enhanced STAT3 phosphorylation, MCP-1 expression, and neointima formation. In addition, whereas dominant negative Src blocked BI-induced 15-Lox1-enhanced Jak2 phosphorylation, dnJak2 had no effect on Src phosphorylation. Together, these observations demonstrate for the first time that the 15-Lox1-15(S)-HETE axis activates EGFR via redox-sensitive manner, which in turn mediates Src-Jak2-STAT3-dependent MCP-1 expression leading to vascular wall remodeling. | 21536676
 |
JAK2 exon 12 mutations in polycythemia vera and idiopathic erythrocytosis.
Scott, Linda M, et al.
N. Engl. J. Med., 356: 459-68 (2007)
2007
Show Abstract
BACKGROUND: The V617F mutation, which causes the substitution of phenylalanine for valine at position 617 of the Janus kinase (JAK) 2 gene (JAK2), is often present in patients with polycythemia vera, essential thrombocythemia, and idiopathic myelofibrosis. However, the molecular basis of these myeloproliferative disorders in patients without the V617F mutation is unclear. METHODS: We searched for new mutations in members of the JAK and signal transducer and activator of transcription (STAT) gene families in patients with V617F-negative polycythemia vera or idiopathic erythrocytosis. The mutations were characterized biochemically and in a murine model of bone marrow transplantation. RESULTS: We identified four somatic gain-of-function mutations affecting JAK2 exon 12 in 10 V617F-negative patients. Those with a JAK2 exon 12 mutation presented with an isolated erythrocytosis and distinctive bone marrow morphology, and several also had reduced serum erythropoietin levels. Erythroid colonies could be grown from their blood samples in the absence of exogenous erythropoietin. All such erythroid colonies were heterozygous for the mutation, whereas colonies homozygous for the mutation occur in most patients with V617F-positive polycythemia vera. BaF3 cells expressing the murine erythropoietin receptor and also carrying exon 12 mutations could proliferate without added interleukin-3. They also exhibited increased phosphorylation of JAK2 and extracellular regulated kinase 1 and 2, as compared with cells transduced by wild-type JAK2 or V617F JAK2. Three of the exon 12 mutations included a substitution of leucine for lysine at position 539 of JAK2. This mutation resulted in a myeloproliferative phenotype, including erythrocytosis, in a murine model of retroviral bone marrow transplantation. CONCLUSIONS: JAK2 exon 12 mutations define a distinctive myeloproliferative syndrome that affects patients who currently receive a diagnosis of polycythemia vera or idiopathic erythrocytosis. | 17267906
 |