119140 Sigma-AldrichAdenosine Receptor Agonist, NECA - CAS 35920-39-9 - Calbiochem
Recommended Products
Overview
| Replacement Information |
|---|
Pricing & Availability
| Catalog Number | Availability | Packaging | Qty/Pack | Price | Quantity | |
|---|---|---|---|---|---|---|
| 119140-10MG |
|
Glass bottle | 10 mg |
|
— |
| Product Information | |
|---|---|
| CAS number | 35920-39-9 |
| Form | White solid |
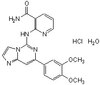
| Hill Formula | C₁₂H₁₆N₆O₄ |
| Chemical formula | C₁₂H₁₆N₆O₄ |
| Reversible | Y |
| Structure formula Image | |
| Quality Level | MQ100 |
| Applications |
|---|
| Biological Information | |
|---|---|
| Primary Target K<sub>i</sub> | 14 nM |
| Purity | ≥98% by HPLC |
| Physicochemical Information |
|---|
| Dimensions |
|---|
| Materials Information |
|---|
| Toxicological Information |
|---|
| Safety Information according to GHS |
|---|
| Safety Information |
|---|
| Product Usage Statements |
|---|
| Packaging Information | |
|---|---|
| Packaged under inert gas | Packaged under inert gas |
| Transport Information |
|---|
| Supplemental Information |
|---|
| Specifications |
|---|
| Global Trade Item Number | |
|---|---|
| Catalog Number | GTIN |
| 119140-10MG | 04055977223354 |
Documentation
Adenosine Receptor Agonist, NECA - CAS 35920-39-9 - Calbiochem SDS
| Title |
|---|
Adenosine Receptor Agonist, NECA - CAS 35920-39-9 - Calbiochem Certificates of Analysis
| Title | Lot Number |
|---|---|
| 119140 |
References
| Reference overview |
|---|
| Forster, K., et al. 2006. Basic Res. Cardiol. 101, 319. de Zwart, M., et al. 1998. Nucleosides Nucleotides. 17, 969. Phillis, J. et al. 1986, Pharm. Biochem. Behavior.24, 263 Durcan, M. J., et al. 1989. Pharmacol Biochem Behav. 32, 487. Bacher, S., et al. 1982. Naunyn Schmiedebergs Arch Pharmacol. 329, 67. Cusack, N., et al. 1981. Br J Pharm.72, 443. Londos, C., et al. 1980. Proc Natl Acad Sci.77, 2551. |