Curcumin attenuates brain edema in mice with intracerebral hemorrhage through inhibition of AQP4 and AQP9 expression.
Wang, BF; Cui, ZW; Zhong, ZH; Sun, YH; Sun, QF; Yang, GY; Bian, LG
Acta pharmacologica Sinica
36
939-48
2015
Show Abstract
Aquaporins (AQPs) are the water-channels that play important roles in brain water homeostasis and in cerebral edema induced by brain injury. In this study we investigated the relationship between AQPs and a neuroprotective agent curcumin that was effective in the treatment of brain edema in mice with intracerebral hemorrhage (ICH).ICH was induced in mice by autologous blood infusion. The mice immediately received curcumin (75, 150, 300 mg/kg, ip). The Rotarod test scores, brain water content and brain expression of AQPs were measured post ICH. Cultured primary mouse astrocytes were used for in vitro experiments. The expression of AQP1, AQP4 and AQP9 and NF-κB p65 were detected using Western blotting or immunochemistry staining.Curcumin administration dose-dependently reduced the cerebral edema at d 3 post ICH, and significantly attenuated the neurological deficits at d 5 post ICH. Furthermore, curcumin dose-dependently decreased the gene and protein expression of AQP4 and AQP9, but not AQP1 post ICH. Treatment of the cultured astrocytes with Fe(2+) (10-100 μmol/L) dose-dependently increased the expression and nuclear translocation of NF-κB p65 and the expression of AQP4 and AQP9, which were partly blocked by co-treatment with curcumin (20 μmol/L) or the NF-κB inhibitor PDTC (10 μmol/L).Curcumin effectively attenuates brain edema in mice with ICH through inhibition of the NF-κB pathway and subsequently the expression of AQP4 and AQP9. Curcumin may serve as a potential therapeutic agent for ICH. | | | 26119880
 |
Connexin 43 stabilizes astrocytes in a stroke-like milieu to facilitate neuronal recovery.
Wu, LY; Yu, XL; Feng, LY
Acta pharmacologica Sinica
36
928-38
2015
Show Abstract
Connexin 43 (Cx43) is a member of connexin family mainly expressed in astrocytes, which forms gap junctions and hemichannels and maintains the normal shape and function of astrocytes. In this study we investigated the role of Cx43 in astrocytes in facilitating neuronal recovery during ischemic stroke.Primary culture of astrocytes or a mixed culture of astrocytes and cortical neurons was subjected to oxygen glucose deprivation and reperfusion (OGD/R). The expression of Cx43 and Ephrin-A4 in astrocytes was detected using immunocytochemical staining and Western blot assays. Intercellular Ca(2+) concentration was determined with Fluo-4 AM fluorescent staining. Middle cerebral artery occlusion (MCAO) model rats were used for in vivo studies.OGD/R treatment of cultured astrocytes caused a decrement of Cx43 expression and translocation of Cx43 from cell membrane to cytoplasm, accompanied by cell retraction. Furthermore, OGD/R increased intracellular Ca(2+) concentration, activated CaMKII/CREB pathways and upregulated expression of Ephrin-A4 in the astrocytes. All these changes in OGD/R-treated astrocytes were alleviated by overexpression of Cx43. In the cortical neurons cultured with astrocytes, OGD/R inhibited the neurite growth, whereas overexpression of Cx43 or knockdown of Ephrin-A4 in astrocytes restored the neurite growth. In MCAO model rats, neuronal recovery was found to be correlated with the recuperation of Cx43 and Ephrin-A4 in astrocytes.Cx43 can stabilize astrocytes and facilitate the resistance to the deleterious effects of a stroke-like milieu and promote neuronal recovery. | | | 26095039
 |
Expression of progerin in aging mouse brains reveals structural nuclear abnormalities without detectible significant alterations in gene expression, hippocampal stem cells or behavior.
Baek, JH; Schmidt, E; Viceconte, N; Strandgren, C; Pernold, K; Richard, TJ; Van Leeuwen, FW; Dantuma, NP; Damberg, P; Hultenby, K; Ulfhake, B; Mugnaini, E; Rozell, B; Eriksson, M
Human molecular genetics
24
1305-21
2015
Show Abstract
Hutchinson-Gilford progeria syndrome (HGPS) is a segmental progeroid syndrome with multiple features suggestive of premature accelerated aging. Accumulation of progerin is thought to underlie the pathophysiology of HGPS. However, despite ubiquitous expression of lamin A in all differentiated cells, the HGPS mutation results in organ-specific defects. For example, bone and skin are strongly affected by HGPS, while the brain appears to be unaffected. There are no definite explanations as to the variable sensitivity to progeria disease among different organs. In addition, low levels of progerin have also been found in several tissues from normal individuals, but it is not clear if low levels of progerin contribute to the aging of the brain. In an attempt to clarify the origin of this phenomenon, we have developed an inducible transgenic mouse model with expression of the most common HGPS mutation in brain, skin, bone and heart to investigate how the mutation affects these organs. Ultrastructural analysis of neuronal nuclei after 70 weeks of expression of the LMNA c.1824Cgreater than T mutation showed severe distortion with multiple lobulations and irregular extensions. Despite severe distortions in the nuclei of hippocampal neurons of HGPS animals, there were only negligible changes in gene expression after 63 weeks of transgenic expression. Behavioral analysis and neurogenesis assays, following long-term expression of the HGPS mutation, did not reveal significant pathology. Our results suggest that certain tissues are protected from functional deleterious effects of progerin. | | | 25343989
 |
miR-26a and miR-384-5p are required for LTP maintenance and spine enlargement.
Gu, QH; Yu, D; Hu, Z; Liu, X; Yang, Y; Luo, Y; Zhu, J; Li, Z
Nature communications
6
6789
2015
Show Abstract
Long-term potentiation (LTP) is a form of synaptic plasticity that results in enhanced synaptic strength. It is associated with the formation and enlargement of dendritic spines-tiny protrusions accommodating excitatory synapses. Both LTP and spine remodelling are crucial for brain development, cognition and the pathophysiology of neurological disorders. The role of microRNAs (miRNAs) in the maintenance of LTP, however, is not well understood. Using next-generation sequencing to profile miRNA transcriptomes, we demonstrate that miR-26a and miR-384-5p specifically affect the maintenance, but not induction, of LTP and different stages of spine enlargement by regulating the expression of RSK3. Using bioinformatics, we also examine the global effects of miRNA transcriptome changes during LTP on gene expression and cellular activities. This study reveals a novel miRNA-mediated mechanism for gene-specific regulation of translation in LTP, identifies two miRNAs required for long-lasting synaptic and spine plasticity and presents a catalogue of candidate 'LTP miRNAs'. | | | 25858512
 |
Evidence for a novel functional role of astrocytes in the acute homeostatic response to high-fat diet intake in mice.
Buckman, LB; Thompson, MM; Lippert, RN; Blackwell, TS; Yull, FE; Ellacott, KL
Molecular metabolism
4
58-63
2015
Show Abstract
Introduction of a high-fat diet to mice results in a period of voracious feeding, known as hyperphagia, before homeostatic mechanisms prevail to restore energy intake to an isocaloric level. Acute high-fat diet hyperphagia induces astrocyte activation in the rodent hypothalamus, suggesting a potential role of these cells in the homeostatic response to the diet. The objective of this study was to determine physiologic role of astrocytes in the acute homeostatic response to high-fat feeding.We bred a transgenic mouse model with doxycycline-inducible inhibition of NFkappaB (NFκB) signaling in astrocytes to determine the effect of loss of NFκB-mediated astrocyte activation on acute high-fat hyperphagia. ELISA was used to measure the levels of markers of astrocyte activation, glial-fibrillary acidic protein (GFAP) and S100B, in the medial basal hypothalamus.Inhibition of NFκB signaling in astrocytes prevented acute high-fat diet-induced astrocyte activation and resulted in a 15% increase in caloric intake (P less than 0.01) in the first 24 h after introduction of the diet.These data reveal a novel homeostatic role for astrocytes in the acute physiologic regulation of food intake in response to high-fat feeding. | | | 25685690
 |
Effect of thymic stimulation of CD4+ T cell expansion on disease onset and progression in mutant SOD1 mice.
Sheean, RK; Weston, RH; Perera, ND; D'Amico, A; Nutt, SL; Turner, BJ
Journal of neuroinflammation
12
40
2015
Show Abstract
The peripheral immune system is implicated in modulating microglial activation, neurodegeneration and disease progression in amyotrophic lateral sclerosis (ALS). Specifically, there is reduced thymic function and regulatory T cell (Treg) number in ALS patients and mutant superoxide dismutase 1 (SOD1) mice, while passive transfer of Tregs ameliorates disease in mutant SOD1 mice. Here, we assessed the effects of augmenting endogenous CD4+ T cell number by stimulating the thymus using surgical castration on the phenotype of transgenic SOD1(G93A) mice.Male SOD1(G93A) mice were castrated or sham operated, and weight loss, disease onset and progression were examined. Thymus atrophy and blood CD4+, CD8+ and CD4+ FoxP3+ T cell numbers were determined by fluorescence activated cell sorting (FACS). Motor neuron counts, glial cell activation and androgen receptor (AR) expression in the spinal cord were investigated using immunohistochemistry and Western blotting. Differences between castrated and sham mice were analysed using an unpaired t test or one-way ANOVA.Castration significantly increased thymus weight and total CD4+ T cell numbers in SOD1(G93A) mice, although Tregs levels were not affected. Despite this, disease onset and progression were similar in castrated and sham SOD1(G93A) mice. Castration did not affect motor neuron loss or astrocytic activation in spinal cords of SOD1(G93A) mice; however, microglial activation was reduced, specifically M1 microglia. We also show that AR is principally expressed in spinal motor neurons and progressively downregulated in spinal cords of SOD1(G93A) mice from disease onset which is further enhanced by castration.These results demonstrate that increasing thymic function and CD4+ T cell number by castration confers no clinical benefit in mutant SOD1 mice, which may reflect an inability to stimulate neuroprotective Tregs. Nonetheless, castration decreases M1 microglial activation in the spinal cord without any clinical improvement and motor neuron rescue, in contrast to other approaches to suppress microglia in mutant SOD1 mice. Lastly, diminished AR expression in spinal motor neurons, which links to another motor neuron disorder, spinal bulbar muscular atrophy (SBMA), may contribute to ALS pathogenesis and suggests a common disease pathway in ALS and SBMA mediated by disruption of AR signalling in motor neurons. | | | 25889790
 |
Dysregulation of astrocyte extracellular signaling in Costello syndrome.
Krencik, R; Hokanson, KC; Narayan, AR; Dvornik, J; Rooney, GE; Rauen, KA; Weiss, LA; Rowitch, DH; Ullian, EM
Science translational medicine
7
286ra66
2015
Show Abstract
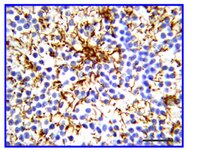
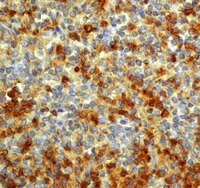
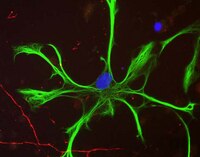
Astrocytes produce an assortment of signals that promote neuronal maturation according to a precise developmental timeline. Is this orchestrated timing and signaling altered in human neurodevelopmental disorders? To address this question, the astroglial lineage was investigated in two model systems of a developmental disorder with intellectual disability caused by mutant Harvey rat sarcoma viral oncogene homolog (HRAS) termed Costello syndrome: mutant HRAS human induced pluripotent stem cells (iPSCs) and transgenic mice. Human iPSCs derived from patients with Costello syndrome differentiated to astroglia more rapidly in vitro than those derived from wild-type cell lines with normal HRAS, exhibited hyperplasia, and also generated an abundance of extracellular matrix remodeling factors and proteoglycans. Acute treatment with a farnesyl transferase inhibitor and knockdown of the transcription factor SNAI2 reduced expression of several proteoglycans in Costello syndrome iPSC-derived astrocytes. Similarly, mice in which mutant HRAS was expressed selectively in astrocytes exhibited experience-independent increased accumulation of perineuronal net proteoglycans in cortex, as well as increased parvalbumin expression in interneurons, when compared to wild-type mice. Our data indicate that astrocytes expressing mutant HRAS dysregulate cortical maturation during development as shown by abnormal extracellular matrix remodeling and implicate excessive astrocyte-to-neuron signaling as a possible drug target for treating mental impairment and enhancing neuroplasticity. | Western Blotting | | 25947161
 |
Characterization of glioma stem-like cells from human glioblastomas.
Yamamuro, S; Okamoto, Y; Sano, E; Ochiai, Y; Ogino, A; Ohta, T; Hara, H; Ueda, T; Nakayama, T; Yoshino, A; Katayama, Y
International journal of oncology
47
91-6
2015
Show Abstract
Glioma stem-like cells (GSCs) could have potential for tumorigenesis, treatment resistance, and tumor recurrence (GSC hypothesis). However, the mechanisms underlying such potential has remained elusive and few ultrastructural features of the cells have been reported in detail. We therefore undertook observations of the antigenic characteristics and ultrastructural features of GSCs isolated from human glioblastomas. Tumor spheres formed by variable numbers of cells, exhibiting a variable appearance in both their size and shape, were frequently seen in GSCs expressing the stem cell surface markers CD133 and CD15. Increased cell nucleus atypia, mitochondria, rough endoplasmic reticulum, coated vesicles, and microvilli, were noted in the GSCs. Furthermore, cells at division phases and different phases of the apoptotic process were occasionally observed. These findings could imply that GSCs have certain relations with human neural stem cells (NSCs) but are primitively different from undifferentiated NSCs. The data may provide support for the GSC hypothesis, and also facilitate the establishment of future glioblastoma treatments targeting GSCs. | | | 25955568
 |
Cuprizone-induced demyelination and demyelination-associated inflammation result in different proton magnetic resonance metabolite spectra.
Praet, J; Orije, J; Kara, F; Guglielmetti, C; Santermans, E; Daans, J; Hens, N; Verhoye, M; Berneman, Z; Ponsaerts, P; Van der Linden, A
NMR in biomedicine
28
505-13
2015
Show Abstract
Conventional MRI is frequently used during the diagnosis of multiple sclerosis but provides only little additional pathological information. Proton MRS ((1) H-MRS), however, provides biochemical information on the lesion pathology by visualization of a spectrum of metabolites. In this study we aimed to better understand the changes in metabolite concentrations following demyelination of the white matter. Therefore, we used the cuprizone model, a well-established mouse model to mimic type III human multiple sclerosis demyelinating lesions. First, we identified CX3 CL1/CX3 CR1 signaling as a major regulator of microglial activity in the cuprizone mouse model. Compared with control groups (heterozygous CX3 CR1(+/-) C57BL/6 mice and wild type CX3 CR1(+/+) C57BL/6 mice), microgliosis, astrogliosis, oligodendrocyte cell death and demyelination were shown to be highly reduced or absent in CX3 CR1(-/-) C57BL/6 mice. Second, we show that (1) H-MRS metabolite spectra are different when comparing cuprizone-treated CX3 CR1(-/-) mice showing mild demyelination with cuprizone-treated CX3 CR1(+/+) mice showing severe demyelination and demyelination-associated inflammation. Following cuprizone treatment, CX3 CR1(+/+) mice show a decrease in the Glu, tCho and tNAA concentrations as well as an increased Tau concentration. In contrast, following cuprizone treatment CX3 CR1(-/-) mice only showed a decrease in tCho and tNAA concentrations. Therefore, (1) H-MRS might possibly allow us to discriminate demyelination from demyelination-associated inflammation via changes in Tau and Glu concentration. In addition, the observed decrease in tCho concentration in cuprizone-induced demyelinating lesions should be further explored as a possible diagnostic tool for the early identification of human MS type III lesions. | | | 25802215
 |
The clinical heterogeneity of coenzyme Q10 deficiency results from genotypic differences in the Coq9 gene
Marta Luna-Sánchez 1 , Elena Díaz-Casado 1 , Emanuele Barca 2 , Miguel Ángel Tejada 3 , Ángeles Montilla-García 3 , Enrique Javier Cobos 3 , Germaine Escames 1 , Dario Acuña-Castroviejo 1 , Catarina M Quinzii 2 , Luis Carlos López
EMBO Mol Med
7(5)
670-87
2015
Show Abstract
Primary coenzyme Q10 (CoQ10) deficiency is due to mutations in genes involved in CoQ biosynthesis. The disease has been associated with five major phenotypes, but a genotype-phenotype correlation is unclear. Here, we compare two mouse models with a genetic modification in Coq9 gene (Coq9(Q95X) and Coq9(R239X)), and their responses to 2,4-dihydroxybenzoic acid (2,4-diHB). Coq9(R239X) mice manifest severe widespread CoQ deficiency associated with fatal encephalomyopathy and respond to 2,4-diHB increasing CoQ levels. In contrast, Coq9(Q95X) mice exhibit mild CoQ deficiency manifesting with reduction in CI+III activity and mitochondrial respiration in skeletal muscle, and late-onset mild mitochondrial myopathy, which does not respond to 2,4-diHB. We show that these differences are due to the levels of COQ biosynthetic proteins, suggesting that the presence of a truncated version of COQ9 protein in Coq9(R239X) mice destabilizes the CoQ multiprotein complex. Our study points out the importance of the multiprotein complex for CoQ biosynthesis in mammals, which may provide new insights to understand the genotype-phenotype heterogeneity associated with human CoQ deficiency and may have a potential impact on the treatment of this mitochondrial disorder. | | | 25802402
 |