Intracellular processing of disease-associated α-synuclein in the human brain suggests prion-like cell-to-cell spread.
Kovacs, GG; Breydo, L; Green, R; Kis, V; Puska, G; Lőrincz, P; Perju-Dumbrava, L; Giera, R; Pirker, W; Lutz, M; Lachmann, I; Budka, H; Uversky, VN; Molnár, K; László, L
Neurobiology of disease
69
76-92
2014
Show Abstract
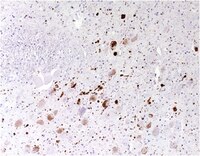
Dementia with Lewy bodies (DLB), Parkinson's disease (PD) and multiple system atrophy are characterized by the deposition of disease-associated α-synuclein. In the present study we 1) examined the molecular specificity of the novel anti-α-synuclein 5G4 antibody; 2) evaluated immunoreactivity patterns and their correlation in human brain tissue with micro- and astrogliosis in 57 cases with PD or DLB; and 3) performed a systematic immunoelectron microscopical mapping of subcellular localizations. 5G4 strongly binds to the high molecular weight fraction of β-sheet rich oligomers, while no binding to primarily disordered oligomers or monomers was observed. We show novel localizations of disease-associated α-synuclein including perivascular macrophages, ependyma and cranial nerves. α-Synuclein immunoreactive neuropil dots and thin threads associate more with glial reaction than Lewy bodies alone. Astrocytic α-synuclein is an important component of the pathology. Furthermore, we document ultrastructurally the pathway of processing of disease-associated α-synuclein within neurons and astroglial cells. Interaction of mitochondria and disease-associated α-synuclein plays a key role in the molecular-structural cytopathogenesis of disorders with Lewy bodies. We conclude that 1) the 5G4 antibody has strong selectivity for β-sheet rich α-synuclein oligomers; 2) Lewy bodies themselves are not the most relevant morphological substrate that evokes tissue lesioning; 3) both neurons and astrocytes internalize disease-associated α-synuclein in the human brain, suggesting prion-like cell-to-cell spread of α-synuclein by uptake from surrounding structures, as shown previously in experimental observations. | 24878508
 |
Clinical neuropathology
Unterberger, U; Lachmann, I; Voigtländer, T; Pirker, W; Berghoff, AS; Flach, K; Wagner, U; Geneste, A; Perret-Liaudet, A; Kovacs, GG
Clinical neuropathology
33
329-34
2014
Show Abstract
With the aim to evaluate the significance and reliability of detecting disease-specific α-synuclein in the cerebrospinal fluid (CSF) we developed an ELISA and bead-assay. We used a commercial antibody (5G4) that does not bind to the physiological monomeric form of α-synuclein, but is highly specific for the disease-associated forms, including high molecular weight fraction of β-sheet rich oligomers. We applied both tests in CSF from a series of neuropathologically confirmed α-synucleinopathy cases, including Parkinson' disease dementia (PDD) and dementia with Lewy bodies (DLB) (n = 7), as well as Alzheimer' disease (n = 6), and control patients without neurodegenerative pathologies (n = 9). Disease-specific α-synuclein was detectable in the CSF in a subset of patients with α-synuclein pathology in the brain. When combined with the analysis of total α-synuclein, the bead-assay for disease-specific α-synuclein was highly specific for PDD/DLB. Detection of disease-associated αsynuclein combined with the total levels of α-synuclein is a promising tool for the in-vivo diagnosis of α-synucleinopathies, including PDD and LBD. | 25131945
 |
In vivo markers of Parkinson's disease and dementia with Lewy bodies: current value of the 5G4 α-synuclein antibody.
Maetzler, W; Pilotto, A; Apel, A; Deuschle, C; Kuebart, G; Heinzel, S; Liepelt-Scarfone, I; Schulte, C; Reusch, D; Schleicher, E; Rothfuss, O; Schneider, A; Dodel, R; Gasser, T; Berg, D
Acta neuropathologica
128
893-5
2014
| 25376537
 |
An antibody with high reactivity for disease-associated α-synuclein reveals extensive brain pathology.
Kovacs, Gabor G, et al.
Acta Neuropathol., 124: 37-50 (2012)
2012
Show Abstract
α-Synuclein is the major protein associated with Lewy body dementia, Parkinson's disease and multiple system atrophy. Since α-synuclein is present in the brain in physiological conditions as a presynaptic protein, it is crucial to characterize disease-associated modifications to develop an in vivo biomarker. With the aim to develop antibodies showing high specificity and sensitivity for disease-associated α-synuclein, synthetic peptides containing different amino acid sequences were used for immunization of mice. After generation of α-synuclein aggregates, ELISA and immunoblotting were used to test the specificity of antibodies. Tissue microarray sections originating from different human α-synucleinopathies were used to compare immunostaining with other, commercially available antibodies. Immunization of mice with the peptide TKEGVVHGVATVAE (amino acid 44-57 of α-synuclein) resulted in the generation of a monoclonal antibody (5G4), which was able to bind aggregated α-synuclein preparation in sandwich ELISA or coated on magnetic beads. 5G4 proved to be superior to other antibodies in comparative immunohistochemical studies by revealing more widespread and distinct α-synuclein pathology. Immunoblotting of human brain tissue revealed an additional band seen in dementia with Lewy bodies, whereas the band representing monomeric α-synuclein was very weak or lacking. In summary, the 5G4 antibody is most promising for re-evaluation of archival material and may offer new perspective for the development of in vivo diagnostic assays for detecting disease-associated α-synuclein in body fluids. | 22370907
 |